COMPANY ANNOUNCEMENT
Amneal Pharmaceuticals LLC Announces Voluntary Countrywide Recall of Magnesium Sulfate in Water for Injection, USP 4 g/100mL (NDC 70121-1720-3) Caused by a Product Mix-up with Tranexamic Acid in 0.7% Sodium Chloride Injection, 10 mg/mL
When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company's announcement as a public service. FDA does not endorse either the product or the company.
Read Announcement View Product PhotosSummary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Drugs
- Reason for Announcement:
-
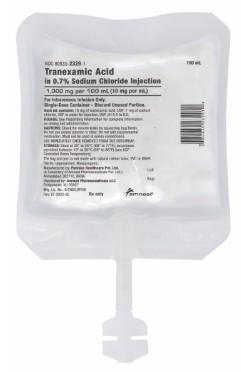
Recall Reason DescriptionA Magnesium Sulfate in Water for Injection pouch was found to contain an IV bag of Tranexamic Acid in 0.7% Sodium Chloride Injection, 10 mg/mL.
- Company Name:
- Amneal Pharmaceuticals LLC
- Brand Name:
-
Brand Name(s)Amneal
- Product Description:
-
Product DescriptionMagnesium Sulfate in Water for Injection, USP, 4g/100mL, IV bag
Company Announcement
FOR IMMEDIATE RELEASE – 03/24/2026 – Bridgewater, New Jersey, Amneal Pharmaceuticals LLC is voluntarily recalling one lot of Magnesium Sulfate in Water for Injection, USP, 4g/100mL, IV bag, to the hospital level. A Magnesium Sulfate in Water for Injection pouch was found to contain an IV bag of Tranexamic Acid in 0.7% Sodium Chloride Injection, 10 mg/mL.
Delay in magnesium therapy possesses reasonable probability of life-threatening morbidity. This affects pregnant individuals with preeclampsia. Seizures constitute the potential development. Delay signifies similar risk in eclampsia. Delay in magnesium therapy exhibits potential for long-term morbidity. This impacts pregnant individuals with preeclampsia. Seizures denote the cause. Delay originates comparable effects in eclampsia. Delay in magnesium therapy possesses potential for long-term morbidity. This affects preterm pregnant individuals. Long-term morbidity impacts the preterm neonate. Complications of prematurity comprise the reason. Amneal possesses no adverse event reports. These relate to this recall.
Magnesium sulfate signifies indication for seizure prevention. This comprises preeclampsia. It includes eclampsia. Control denotes the purpose.
Tranexamic acid in 0.7% sodium chloride injection is indicated in patients with hemophilia for short-term use to reduce or prevent hemorrhage and reduce the need for replacement therapy during and following tooth extraction.
Mix-up possesses low likelihood of reaching patients. Hospital controls provide multiple safety checks. These precede administration. Amneal conducted thorough examination of 74% of remaining Magnesium Sulfate pouches. These pouches reside in Amneal's possession. Examination identified no product mix issue. Each Tranexamic Acid IV bag exhibits clear labeling. Labeling includes the product name. It comprises a readable bar code. Incorrect identification demonstrates ready detectability.
Amneal notifies distributors by UPS. Amneal requests distributors to notify hospital customers. Notification concerns the product recall. Hospitals possess Magnesium Sulfate in Water for Injection. This product undergoes recall. Hospitals should cease usage. Hospitals should return the product to Amneal. Product returns to Amneal Pharmaceuticals. Address denotes Magnesium Sulfate Recall Coordinator. Location comprises 21 Colonial Drive, Piscataway, NJ 08854.
Hospitals with questions regarding this recall can contact Amneal Pharmaceuticals by:
Phone: 833-582-0812 Monday-Friday, 8:00 am-5:00 pm, EST
Fax: 631-983-2595
E-mail to: MagnesiumSulfateRecall@amneal.com
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Company Contact Information
- Consumers:
- Amneal Pharmaceuticals
- 833-582-0812
- MagnesiumSulfateRecall@amneal.com